 Iota-carrageenan formulated into a nasal spray has proven to be safe and effective against coronavirus virus causing common cold. Therefore, the use of antiviral nasal sprays may help reduce nasal and nasopharyngeal viral load, thereby slowing down disease progression and transmission. Furthermore, in the early stage of pathogenesis, the virus is localized mainly in the nasal cavity and the nasopharynx. Recent data have shown that a high viral load and a long virus-shedding period were associated with severe COVID-19. 
Therefore, effective approaches are urgently needed to reduce the spread of the virus and its death toll. However, by the time of this publication, only 3.1% of people in low-income countries have received at least one dose. Ĭurrently, there are no adequate therapeutic or preventive medicines available for COVID-19, except for vaccines. Children are also affected but display milder symptoms than adults, nonetheless, they remain active transmitters of COVID-19. Older adults and those with pre-existing conditions are most susceptible to adverse outcomes. This includes 5% requiring admission to an intensive care unit (ICU), and among these cases, half result in death. However, 15% of patients develop severe pneumonia that requires hospitalization and oxygen support. In most cases, COVID-19 manifests with flu-like symptoms and results in manageable symptoms that resolve without intervention. SARS-CoV-2 is the single-stranded positive-sense RNA virus responsible for COVID-19, causing one of the most significant pandemics of our time, with more than 244,980,203 confirmed cases and more than 4,971,409 deaths worldwide as of October 27 th, 2021. This does not alter our adherence to PLOS ONE’s policies on sharing data and materials. 11.013.687 “PREVENTIVE AND THERAPEUTIC TREATMENT FOR COVID 19 AND ANY OTHER DISEASE CAUSED BY SARS COV 2”, whose inventor is Julio César Vega. This submission is related to US Patent No. The specific roles of these authors are articulated in the ‘Author Contributions’ section.Ĭompeting interests: I have read the journal’s policy and the authors of this manuscript have the following competing interests: I have a commercial affiliation to Laboratorio Pablo Cassará S.R.L, who provided the formulations for the study and to Amcyte Pharma Inc. JMF, AD and CP receive funding from Fundación Pablo Cassará. As disclosed previously, JCV has a commercial affiliation to Amcyte Pharma Inc., as well. JCV receives a salary from Laboratorio Pablo Cassará S.R.L. Samples for this study were prepared by personnel from Laboratorio Pablo Cassará S.R.L. JCV has a commercial affiliation to Amcyte Pharma Inc and has contributed to data analysis and preparation of the manuscript. funded the study and decided to publish it, but did not have any additional role in the study design or, data collection. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.ĭata Availability: All relevant data are within the manuscript and in the supporting information.įunding: Amcyte Pharma Inc. 
Received: JAccepted: NovemPublished: November 19, 2021Ĭopyright: © 2021 Bansal et al. Polyak, University of Washington, UNITED STATES 
(2021) Iota-carrageenan and xylitol inhibit SARS-CoV-2 in Vero cell culture. 329 mM) was found to exert some antiviral action, though this preliminary finding needs further confirmation.Ĭitation: Bansal S, Jonsson CB, Taylor SL, Figueroa JM, Dugour AV, Palacios C, et al. Further, xylitol at a concentration of 50 mg/mL (ca. five times the risk of infection by SARS-CoV-2 in health care personnel. Recently a double-blind, placebo-controlled study showed that iota-carrageenan in isotonic sodium chloride reduces ca. The concentrations of iota-carrageenan with activity against SARS-CoV-2 in vitro may be easily achieved through the application of nasal sprays as commonly used in several countries. 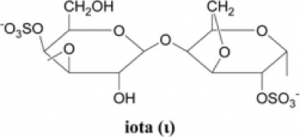
We determined that iota-carrageenan in concentrations as low as 6 μg/mL inhibits SARS-CoV-2 in vitro. In this study, we tested the antiviral action of three candidate nasal spray formulations against SARS-CoV-2 in vitro. Since the nasal cavity and the rhinopharynx are the sites of initial replication of SARS-CoV-2, a nasal spray may be an effective option to target SARS-CoV-2 infection. There is an urgent unmet need to provide an easily producible and affordable medicine to prevent transmission and provide early treatment for this disease. Last year observed a global pandemic caused by SARS-CoV-2 (severe acute respiratory syndrome-coronavirus 2) infection affecting millions of individuals worldwide. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
